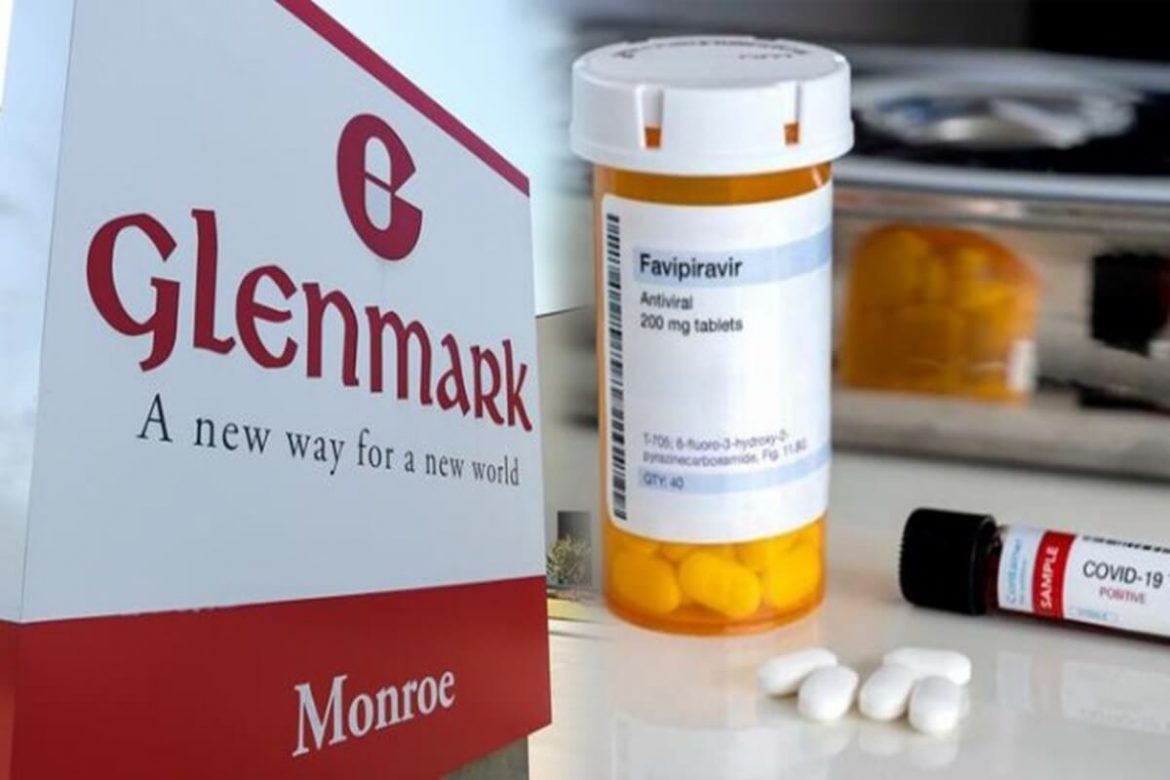
Glenmark Pharmaceuticals on Tuesday said it has initiated Phase 3 clinical trials on antiviral drug Favipiravir to check its efficacy on COVID-19 patients in India.
Mumbai-based Glenmark said in a BSE filing the approval made it the first pharmaceutical company in India to be given the nod by the Drug Controller General of India to start trials on Covid-19 patients.
Favipiravir is manufactured under the brand name Avigan by Fujifilm Toyama Chemical Co Ltd, a subsidiary of Japan’s Fujifilm Holdings Corporation, and approved for use as an anti-flu drug in the country in 2014.
Glenmark said clinical trials have begun and more than 10 leading government and private hospitals in India are being enrolled for the study. It estimates the study completion by July or August this year.
The company said it has successfully developed API and the formulations for the product through its in house research and development team.Several health and medical experts, both in and outside of Glenmark, are eager to see the effect that Favipiravir has on COVID-19 cases.
👉 Click here to read the latest Gujarat news on TheLiveAhmedabad.com